RgAra51A (exo-α-Arabinosidase,AbfA)
RgAra51A (AbfA)
Exo-Ara0314
(EC. 3.2.1.55) exo-α-Arabinosidase
CAZy Family: GH51
PROPERTIES
1. ELECTROPHORETIC PURITY
-Single band on SDS-gel electrophoresis (MW ~56 kDa)
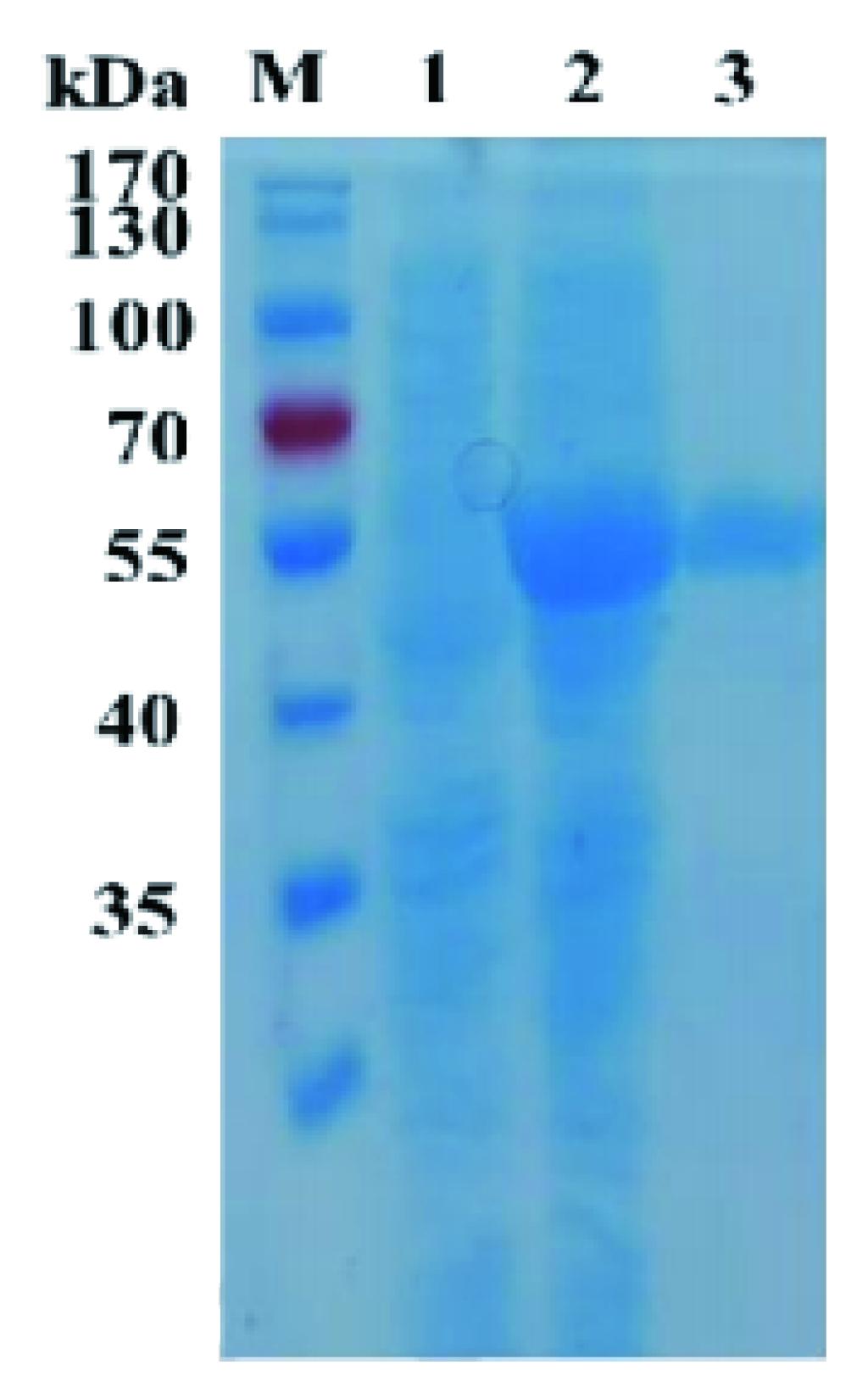
Figure 1. Electrophoresis analysis of RgAra51A. M, molecular weight marker (PageRuler Prestained Protein Ladder, Thermo Scientific); lane 1, culture lysate before IPTG induction; lane 2, culture lysat after IPTG induction; lane 3, RgAra51A purified from Ni sepharose fastflow column.
2. SPECIFIC ACTIVITY
56 U/mg protein (on pNP-α-Araf) at pH 7.0 and 37℃
One Unit of pNP-α-Araf activity is defined as the amount of enzyme required to release 1 μmol of p-nitrophenyl per minute from pNP-α-Araf (5 mM) in phosphate buffer (20 mM) pH 7.0.
3. RELATIVE RATES OF HYDROLYSIS OF SUBSTRATES
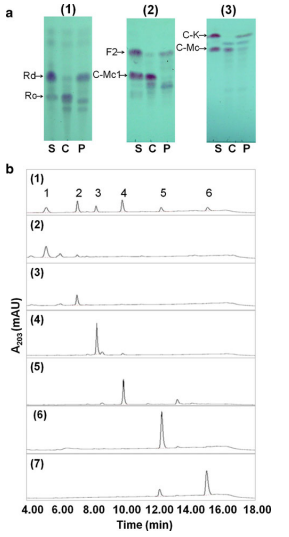
Figure 2. TLC (a) and HPLC (b) analysis of ginsenoside hydrolysis by RgAra51A. a Ginsenoside substrates Rc (1), C-Mc1 (2), and C-Mc (3). S, ginsenoside standard; C, no enzyme; P, reacted with RgAra51A. b Ginsenosides standard (1), Rc (as a reaction control) (2), C-Mc1 (4), and C-Mc (6) standards without enzyme reaction and the products of RgAra51A hydrolysis of Rc (3), C-Mc1 (5), and C-Mc (7). Ginsenoside peaks: 1, Rc; 2, Rd; 3, C-Mc1; 4, F2; 5, C-Mc; and 6, C-K
4. PHYSICOCHEMICAL PROPERTIES
pH Optima: 7.5
pH Stability: 5.0-10.0
Temperature Optima: 37℃
5. STORAGE CONDITIONS
The enzyme should be stored at -20°C. For assay, this enzyme should be diluted in sodium phosphate (20 mM) pH 7.5. Swirl to mix the enzyme immediately prior to use.
6. REFERENCE
[1] An D S, Cui C H, Sung B H, et al. Characterization of a novel ginsenoside-hydrolyzing α-l-arabinofuranosidase, AbfA, from Rhodanobacter ginsenosidimutans Gsoil 3054 T[J]. Applied microbiology and biotechnology, 2012, 94: 673-682.


