BsGal42B(exo-β-1,4-Galactosidase)
BsGal42B
Ex-Gal0061
(EC.3.2.23)exo-β-1,4-Galactosidase
CAZy Family: GH42
PROPERTIES
1.ELECTROPHORETIC PURITY
-Single band on SDS-gel electrophoresis (MW ~79 kDa)
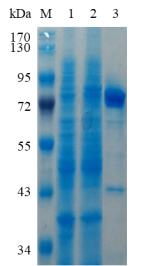
Figure 1. Electrophoresis analysis of BsGal42B. M, molecular weight marker (PageRuler Prestained Protein Ladder, Thermo Scientific); lane 1, culture lysate before IPTG induction; lane 2, culture lysate after IPTG induction; lane 3, BsGal42B purified from Ni sepharose fastflow column.
2.SPECIFIC ACTIVITY
46 U/mg protein (on pNP-β-gal) at pH 7.0 and 40°C
One Unit of pNP-β-gal activity is defined as the amount of enzyme required to release 1 μmol of p-nitrophenyl per minute from pNP-β-gal (5 mM) in phosphate buffer (20 mM) pH 7.0.
3.RELATIVE RATES OF HYDROLYSIS OF SUBSTRATES
Table 1. Relative activity of the purified BsGal42B enzymeon various chromogenic substrates

aActivity on ONPG was taken as 100% and corresponds to a specific activityof 46 U/mg. Results are as measured by oNP or pNP release at 40°C
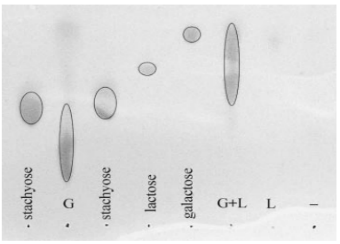
Figure 2.Enzymatic degradation of galactan in vitro. Preparationscontaining either GalA-H (G) or H-LacA (L) were incubated sepa-rately or concurrently (G+L) with galactan for 3 h a t 37°C, and theproducts were analyzed by TLC after precipitation and removal of theremaining whole polysaccharide. A negative control treatment usingno enzymes (-) was also examined. The amount of product applied tothe TLC from the G+L reaction was half that applied from the others(-, L, and G) in order to prevent overloading. Migration patterns ofthe products were compared to those for stachyose, lactose, and ga-lactose.
4.PHYSICOCHEMICAL PROPERTIES
pH Optima:6.5
pH Stability: 6.0-6.5
Temperature Optima:50°C
Temperature Stability: < 50°C
5.STORAGE CONDITIONS
The enzyme should be stored at -20°C. For assay, this enzyme should be diluted in phosphate buffer (20 mM) pH 7.0. Swirl to mix the enzyme immediately prior to use.
6. REFERENCES
[1] Shipkowski S, Brenchley JE. Bioinformatic, genetic, and biochemical evidence that some glycoside hydrolase family 42 beta-galactosidases are arabinogalactan type I oligomer hydrolases. Appl Environ Microbiol. 2006 Dec;72(12):7730-8.


