BhRgl(Rhamnogalacturonan lyase,BhRgl11A)
BhRgl11A
En-Rgl0172
(EC. 4.2.2.23) Rhamnogalacturonan lyase
CAZy Family: PL11
PROPERTIES
1. ELECTROPHORETIC PURITY
-Single band on SDS-gel electrophoresis (MW ~68 kDa)

Figure 1. Electrophoresis analysis of BhRgl11A. M, molecular weight marker (PageRuler Prestained Protein Ladder, Thermo Scientific); lane 1, culture lysate before IPTG induction; lane 2, culture lysate after IPTG induction; lane 3, BhRgl11A purified from Ni sepharose fast flow column.
2. SPECIFIC ACTIVITY
4.9 U/mg protein (on RG-I-AT from Adenophora tetraphylla(Thunb.)Fisch.) at pH8.0 and 30°C.
One unit of RGL activity was defined as the amount of enzyme required to generate 1 μmol of 4,5-unsaturated galacturonic acid in 1 min.
3. RELATIVE RATES OF HYDROLYSIS OF SUBSTRATES
Table 1 Substrate specificity of BhRgl11A
Substrate (0.5 %, w/v) |
Relative activity (%) |
Rhamnogalacturonan-I |
100±0.00 |
Pectin |
62.50±0.48 |
Pectic Galactan |
18.18±0.44 |
Galactan |
- |
Polygalacturonic acid |
- |
4. PHYSICOCHEMICAL PROPERTIES
pH Optima: 8.0
pH Stability: 7.0-9.0
Temperature Optima: 55°C
Temperature Stability: < 50°C

Figure 2. Effect of temperature and pH on the activity and stability of recombinant BhRgl11A. (A) Optimum pH. Assays were carried out at 55°C for 10 min in buffers ranging in pH from 2.0 to 11.0. The activity at optimum pH was defifined as 100%. (B) pH stability. Assays were carried out after the incubation of the enzyme in buffers ranging from pH 2.0-11.0 at 4°C. (C) Optimum temperature. Activity was measured between 20°C and 80°C at pH 8.0 for 10 min. The activity at optimum temperature was defifined as 100%. (D) Thermostability. Assays were carried out in 50 mM phosphate buffer (pH 7.0) at 55°C for 10 min after the incubation of the enzyme at 40-60°C. The activity without heat treatment was defifined as 100%.
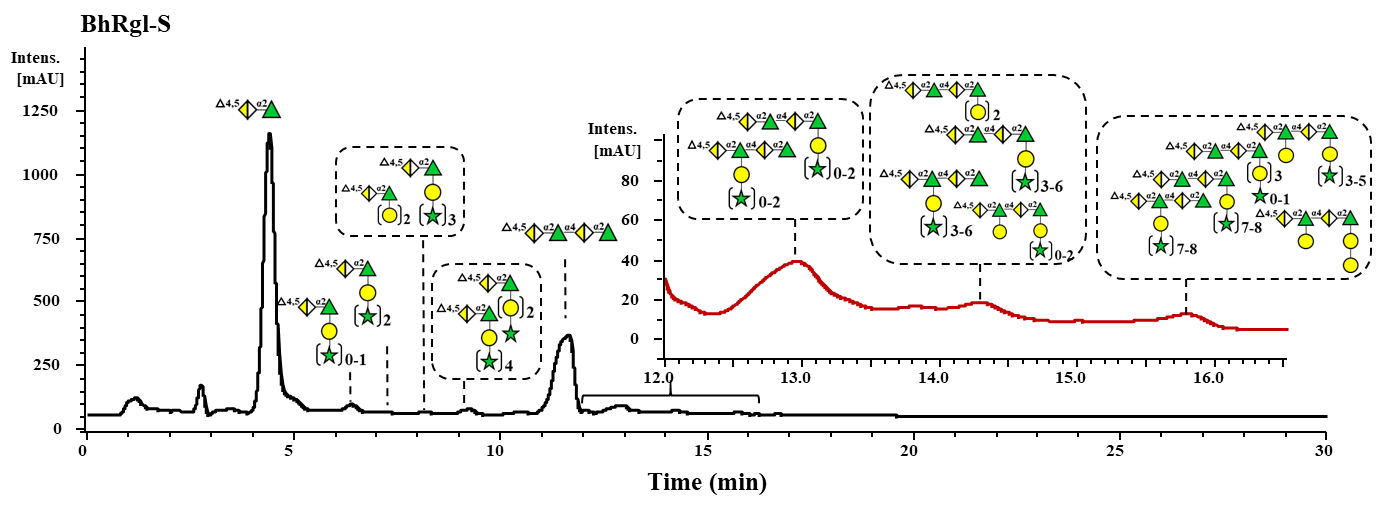
Figure 3. HILIC-UV ( UV235nm ) chromatograms of BhRgl11A oligosaccharides.
5. STORAGE CONDITIONS
The enzyme should be stored at -20°C. For assay, this enzyme should be diluted in phosphate buffer (50 mM) pH 8.0. Swirl to mix the enzyme immediately prior to use.
6. REFERENCE
[1]石慧敏.八种药用植物RG-I 型果胶侧链结构及其分布研究[D]. 东北师范大学, 2021.


