BtAra43A (exo-α-1,2/3-Arabinofuranosidase)
BtAra43A
Ex-Ara0131
(EC.3.2.1.55)exo-α-1,2/3-Arabinofuranosidase
CAZy Family: GH43
PROPERTIES
1.ELECTROPHORETIC PURITY
-Single band on SDS-gel electrophoresis (MW ~38kDa)
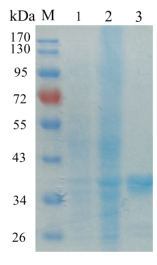
Figure 1. Electrophoresis analysis ofBtAra43A. M, molecular weight marker (PageRuler Prestained Protein Ladder, Thermo Scientific); lane 1, culture lysate before IPTG induction; lane 2, culture lysate after IPTG induction; lane 3, BtAra43A purified from Ni sepharose fastflow column.
2.SPECIFIC ACTIVITY
1.51 U/mg protein (on pNPαGal) at pH 7.0 and 37°C.
0.75 U/mg protein (on pNPαAraf) at pH 7.0 and 37°C.
One Unit of pNPαGal /pNPαAraf activity is defined as the amount of enzyme required to release one μmole of p-nitrophenyl per minute from pNPαGal/pNPαAraf (5 mM) in pH 7 phosphate buffer (20 mM) .
3.RELATIVE RATES OF HYDROLYSIS OF SUBSTRATES
Table 1. Relative activity of BtAra43A on different substratesa.
Substrateb |
Relative activity (%)c |
pNPβGlu |
_ |
pNPβGal |
_ |
pNPβMan |
_ |
pNPβXyl |
_ |
pNPαGlu |
_ |
pNPαGal |
100b±0.0 |
pNPαMan |
_ |
pNPαAraf |
_ |
pNPαArap |
_ |
aReactions were performed with 5 mM substrate, pH 7.0, at 37°C for 5 min.
bAbsorption caused by released p-nitrophenol was measured at 405 nm. The relative activity on pNPαAra was taken as 100%.
cThe data are reported as means±standard errors from the mean for three independent experiments.
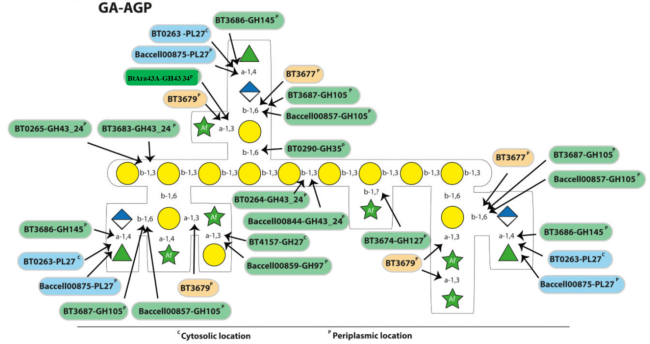
Figure 2. The structure of arabinogalactans, PULs upregulated by the glycans and enzymes that attack these glycans. Structure of gum arabic (GA-AGP) arabinogalactans, and the enzymes that act on these glycans. The enzymes are identified by their locus tag (BTXXXX and BaccellXXXX are derived from B. thetaiotaomicron and B. cellulosilyticus, respectively), assignment to cazy families (GHXX and PLXX indicate glycoside hydrolase and polysaccharide lyase families, respectively) and their predicted cellular location.
4.PHYSICOCHEMICAL PROPERTIES
pH suggestion: 7.0
Temperature suggestion: 37°C
5.STORAGE CONDITIONS
The enzyme should be stored at -20 °C. For assay, this enzyme should be diluted in sodiumphosphate (20 mM) pH 7.0. Swirl to mix the enzyme immediately prior to use.
6. REFERENCES
[1]Cartmell A, Muñoz-Muñoz J, Briggs J A, et al. A surface endogalactanase in Bacteroides thetaiotaomicron confers keystone status for arabinogalactan degradation. Nature microbiology, 2018, 3(11): 1314-1326.


